|
As such, the role of the f orbitals in bonding and reactivity has been a subject of considerable debate. Thus, bonding in the lanthanides and actinides is thought to rely more heavily on the p and d orbitals. The 14 electrons that can reside in these orbitals are highly contracted (i.e., held close to the nucleus) and are not thought to overlap to any great degree with the valence orbitals of neighboring atoms. 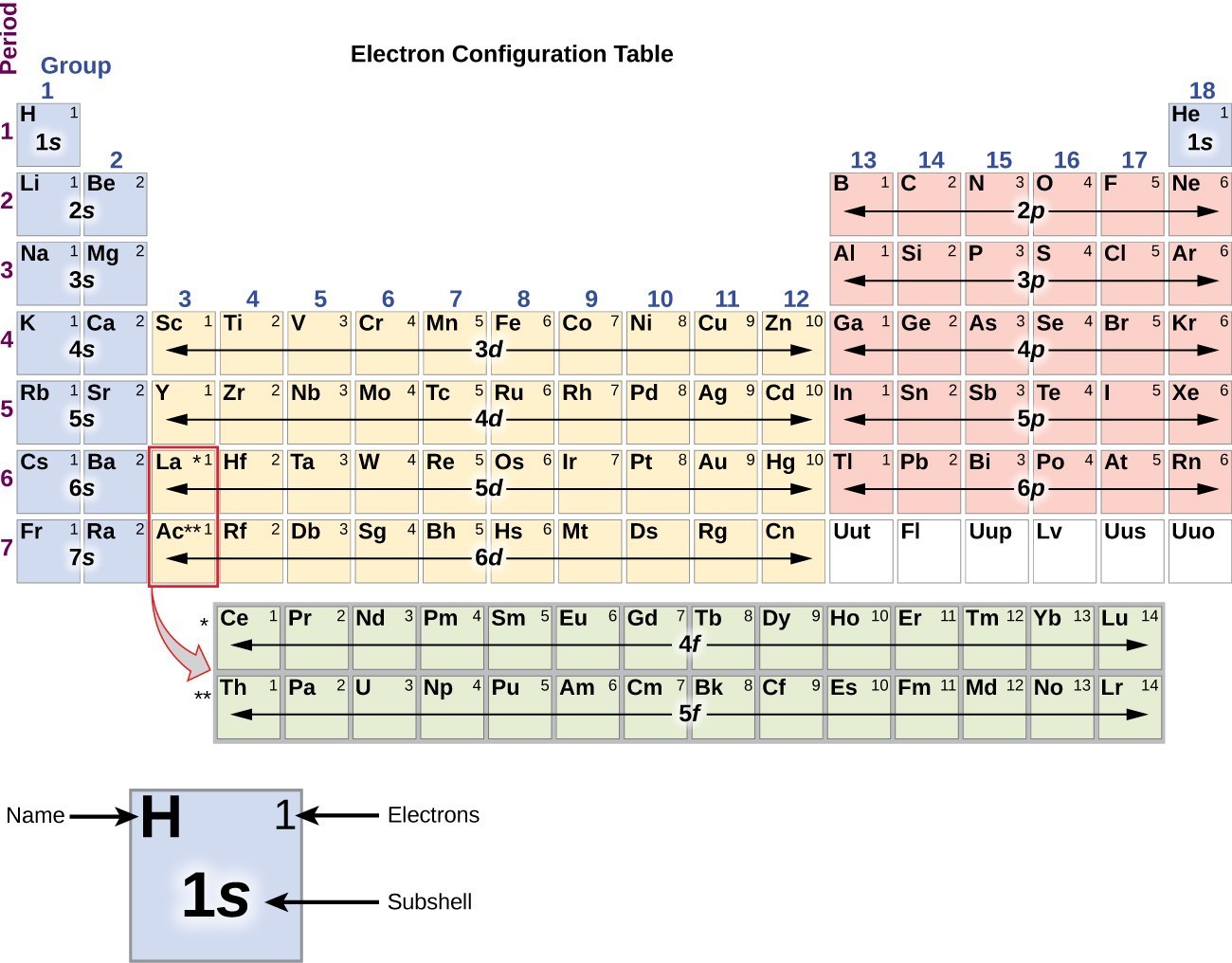
In contrast to the transition elements, the seven f orbitals, which are found in lanthanides and actinides, are less well understood. Thus, electrons in the 3s orbital of sodium (Na) are higher in energy and farther away from the nucleus than electrons found in the 2s orbital of lithium (Li). A larger number indicates a larger and higher energy orbital. This number is an indication of the size and energy of the orbital. Orbitals are often preceded by numerical designations, i.e. These elements use electrons in the d orbitals for bonding and chemical reactivity. For example, elements having a partially filled set of d orbitals are called transition, or d-block, elements. The type of orbital (s, p, d, or f) that the valence electrons reside in is a function of the elements' position in the periodic table. These electrons, called the valence electrons, are the most loosely held and interact with those in other atoms to form chemical bonds. The chemical and physical behavior of the elements results from the configuration of the outermost electrons. The shape and orientation of the d orbitals, which together can hold up to 10 electrons, are shown to the right. 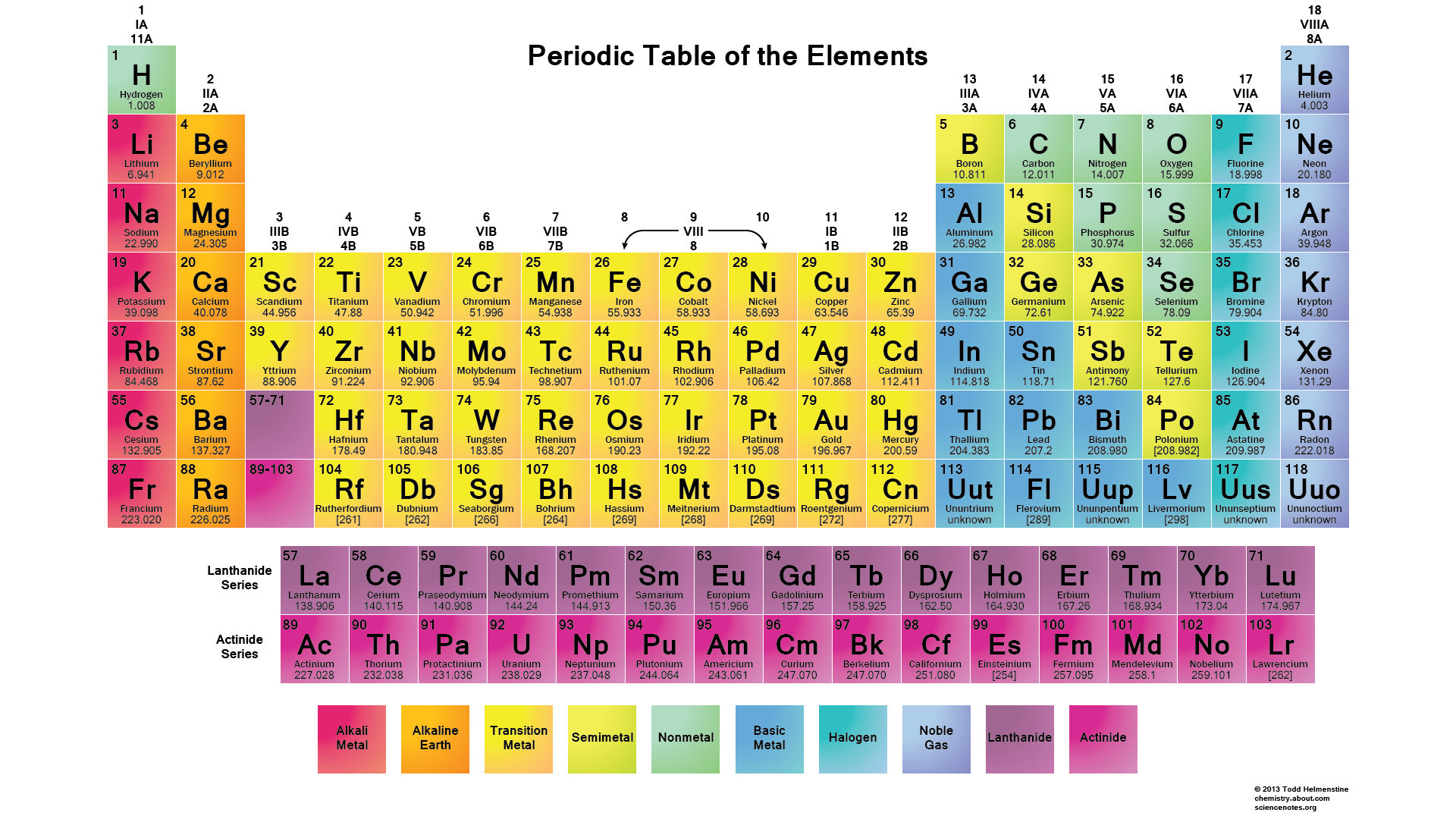
There are five d orbitals, which have more complicated shapes than s and p orbitals. Generally, elements in Groups 1, 2, and 13 to 17 tend to react to form a closed shell, corresponding to the electron configuration s2p6. That is why elements whose atoms have the same number of valence electrons are grouped together in the Periodic Table. The p orbitals can hold up to six electrons. The valence electrons are the electrons in the outermost electron shell of an atom. There are three p orbitals, each of which has the same basic dumbbell shape but differ in its orientation in space. An s orbital has a spherical shape and can hold two electrons. There are four basic types of orbitals: s, p, d, and f. An orbital is a region of space where there is a high probability of finding an electron. Early models of the atom depicted the electrons circling the nucleus in fixed orbits, much like planets revolving around the sun.Ĭurrent theory suggests that electrons are housed in orbitals. Oxygen will gain 2 electrons.The nucleus is the central core of an atom and is made up of protons and neutrons. Electrons are very light, negatively charged particles that surround the positively charged nucleus. The second shell has six electrons ( 2 s 22 p 4) and needs two electrons to achieve octet. The electron configuration of O atom is 1 s 22 s 22 p 4. How many electrons must O lose/gain to achieve octet? Write the formula of the resulting ion and its electron configuration. Write the electron configuration of oxygen atom (Z=8). In 1869 Russian chemist Dmitri Mendeleev. Meanwhile, elements in the same period have the same number of occupied electron shells. Elements in the same group have the same number of valence electrons. In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. The periodic table is organized into groups (vertical columns), periods (horizontal rows), and families (groups of elements that are similar). The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. Notice that there are no leftover electrons. The resulting combination is the compound sodium chloride. With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. On the right, the chloride ion has 18 electrons and has a 1− charge. 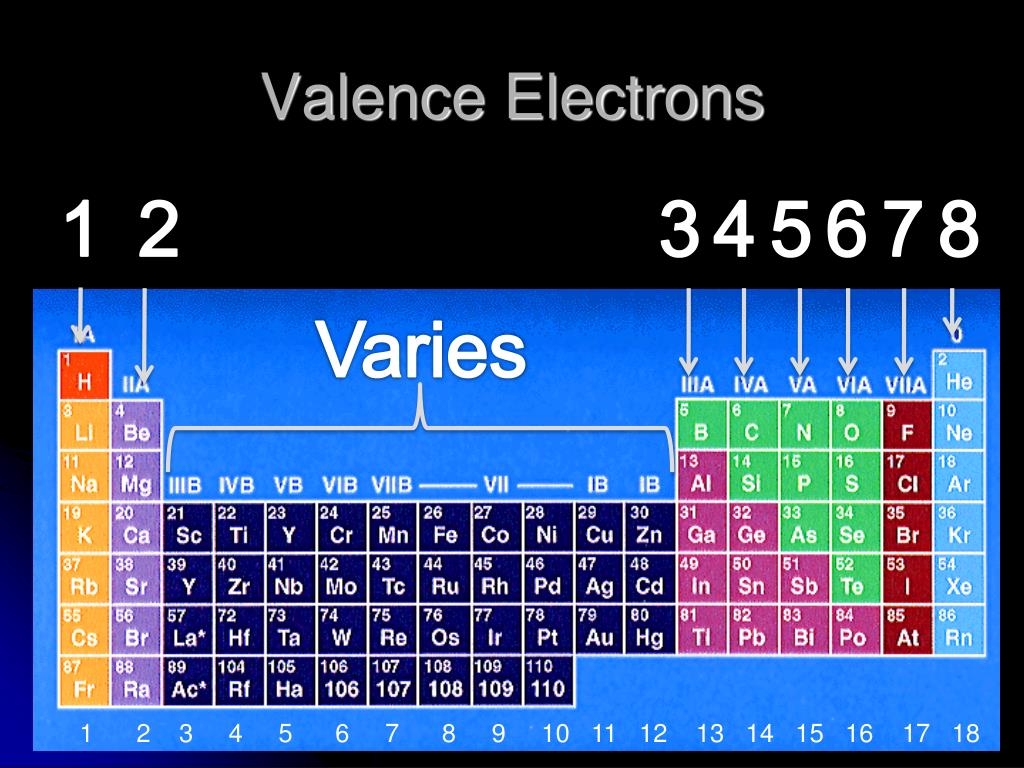
In a multi-electron system, electron penetration is defined by an electrons. Penetration describes the proximity to which an electron can approach to the nucleus. for the elements of the first three rows of the periodic table. On the left, the chlorine atom has 17 electrons. A simple approximation is that all other non-valence electrons shield equally and fully: Si1 label.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
